Posts over on this topic HISO 10099 NZ International Patient Summary (NZIPS) have spilled into an interesting broader discussion on health domain data modelling. For posterity, I’m hoping to lure that conversation to this new heading and I’ll invite the Hira team to post their developing entity model here for comment
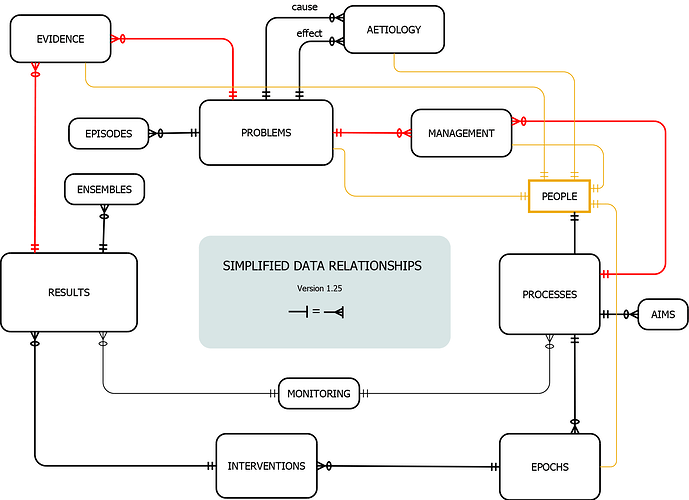
For what it’s worth, here’s the ERD of a clinical data structure that works for me.
Explanation
As it’s cyclical, you can start anywhere, but for convenience (and in terms of the model of science that I’ve previously discussed) let’s start with problems. They can be supported by evidence (even with appropriate Bayesian weighting) that links to results in a many-to-many mapping.
In brief, causal assertions can be made through the aetiology table. Problems also engender the establishment of processes that extend over time (via management, in a similar many-to-may way). The intent associated with a process is further clarified by the AIMS table; an example would be a prescription. (A few years ago I prototyped a fairly comprehensive NZULM-based prescribing system using this model). Clearly, there can be multiple concurrent processes of various types.
Let’s take a specific process like delivery of an anaesthetic. It has a start and an end. It’s possible that multiple actors will play various roles and can start/leave. This is accommodated: each single interaction between patient and caregiver in the context of a process is represented by an epoch. Interventions that take place in this context might be administration of a drug, performing a procedure, or recording of an observation—all of which translate pretty naturally into results. And so the cycle continues.
Note that your issue of multiple ‘streamed’ measurements is well catered for by the ENSEMBLES table attached to results. The idea here is to minimise the amount of memory used for repetitive measures. I’ve put in ten years of data from our SAFERsleep anaesthetic database at ADHB (multiple streams recorded at 20s to 30s intervals) and several hundred thousand full anaesthetics come in at about 60GB, so it’s quite efficient.
This is obviously just a sketch, but a full SQL implementation exists and I’ve used it in various clinical contexts. The details and mapping to coding systems like NZULM, SNOMED CT, NHI, HPI and ICD-10-AM procedural codes become a bit more complex, but not intractably so. A canonical structure for most tables addresses a number of the concerns you’ve expressed, and timestamps in my version are consistently and completely mapped to IANA tz (as proleptic Julian GPS time with high resolution) removing a lot of timestamp/time-zone issues as well as the issue of non-standard timestamps we see with so many RDBMSs. The ERD also doesn’t show a second-tier mapping that accommodates individual usergroups and their representational needs. Practically, the North-West corner also needs some fleshing out.
If you’re interested, happy to provide full source code under the GPL.
It may be best to see this as something to hold other systems to, as I doubt there will be a huge appetite for the structure itself for another 20–50 years. We need to fail conspicuously and painfully first ![]()
Regards, Jo.
Thanks for that.
I buy it on cursory examination (I use a simpler model in my head and want to reconcile them but agree it is circular) and I see your point re: FHIR and I think this helps a lot in terms of that (ie. a strategy).
The problem with FHIR being that it names a bunch of the items in your schema but doesn’t show the systematised/ontological view of the resources. I think you have hit on the kernel of what bothers me the most about IT people and how they design systems - they don’t understand the flow or relationships about why stuff happens in healthcare and the natural order of the process.
Let me have a couple of conversations at my end about this and I will come back to you.
Jon
At first glance, this model looks to be extremely hospital-centric, so it would be interesting to see how it might be applied to community settings.
The fact that the FHIR Resource Model doesn’t contain information model bindings to a specific terminology or ontology , such as SNOMED CT, is deliberate. There are various reasons for this - for example, as those of us who have undertaken the SNOMED International Courses have been taught, terminology and information models often don’t align and even a cursory comparison of the FHIR Resource Model and SNOMED CT hierarchies will support that.
Finally, a slight rant In close to 4 decades in IT, in various domains, I’d venture to say that healthcare has had the greatest input from users - not to mention that everyone in this domain has personal experience of healthcare systems, many of us in different countries. Therefore, to suggest that ‘IT People’ are the problem is fatuous and unhelpful. Among other things, Health Informatics is intended to bridge the game between clinicians and technicians - not an arena where one blames the other.
Apologies for the generalisation @pkjordan it was insensitive and I obviously didn’t mean you.
I do think the issue is credible though that many IT people who have worked in health don’t understand healthcare delivery (I had an example today) but it also affects most of the health people as well (for different reasons). The picture I use to talk about this is this one:
In general health professionals look at this and say - wow what a good idea, IT people generally think it is insane.
I am comfortable that the flow above is reasonably widely applicable, it feels like a pretty common clinical reasoning rubric - the time to go around the process is probably accelerated in an inpatient context but is consistent in the community (social care might be slightly different - but not hugely).
Hope this helps,
Jon
Apology accepted @jon_herries - there are frustrations on both sides and a great deal of scope for increasing mutual understanding. Many clinical workflows appear to be complex and non-standard, and using technology to support them (other than by providing static data) is extremely challenging. Sometimes, the answer is to change those workflows - for example, moving to paperless, signature-exempt prescriptions will make e-Prescribing systems far more efficient than continuing to work with paper scripts in a digital environment.
The picture is interesting, I rather like it! ![]()
I’m really interested in clinical informaticians’ feedback on @DrJo 's model. In a community setting, a litmus test for me is how it might incorporated a shared care plan.
Hi @pkjordan
I agree that there isn’t much point in taking sides and sniping. Co-operation is essential among all participants.
I’m a little puzzled why you consider my model “extremely hospital-centric” and would value your thoughts on what you see (or perceive) that impedes it from being used generally.
What I would value even more is an ERD that illustrates the thinking that underpins HISO 10099, especially if it provides a joined-up model that is superior to my little sketch. When I look at HISO 10099:2022 all I see is entities without relationships.
This does concern me. I remember sitting down about a decade ago for a full day seminar with the Ocean chaps looking at OpenEHR, and my lasting memory was twofold. The first was a lot of semantic confusion about observation | condition | problem | diagnosis. The second was that despite asking for ‘clinician input’ they had a fixed, ontological view of medicine that they were seeking to impose on clinicians—and were frustrated that clinicians were ‘untrainable’ (although perhaps this wasn’t the word they used).
A couple of years ago at HINZ, I sat in a joint FHIR session for programmers and clinicians. Right before my eyes, the two partitioned. The programmers quickly knocked together a prototype system for allergies, and were very proud of this. They became a bit unhappy when the clinicians pointed out that their terminology didn’t fit with the way we described allergies, and that what they had put together was clinically unsuitable. The meeting ended without joy.
Actually, in the above discussion, I pointed out that the same thing still seems to be happening with FHIR and allergies. The example I gave was that the provisional list of options related to Anaphylaxis doesn’t include one of the three most important clinical findings—almost certainly the most important one—hypotension. But this is, of course, just an isolated example. One might think it is easily fixed. I think the problem runs deeper.
There’s a more general problem that I’ve also briefly alluded to. This is ‘hard-coded’ design around complex ontologies. Let’s say that you’ve built part of your structure around say “an anaphylactoid reaction” (and its relationships). We then decide that this is a meaningless term. Now we have a structural problem. Conversely, if the term is merely another entry in a table, it can be flagged as obsolete. But in the absence of a clinically solid ERD, how do we instantiate aspirational documents like HISO 10099? How do we avoid hard-coded, unstable or prolix design ‘features’? And how do we know what are essential features that must be present for us to do good clinical science?
I’d suggest that just letting everyone “get on with their own approach” will multiply our current dysfunction. Yet previously on the FHIR discussion list, this is pretty much the response that I’ve received when I’ve suggested that it’s not enough to have a messaging standard without underpinning it using a joined up model. The message I received was “We’re only concerned with the messaging”. Perhaps I misunderstood this?
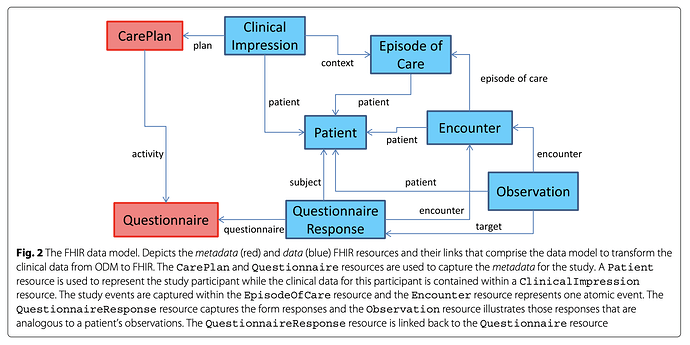
As I’ve already noted, if you want FHIR to provide ‘semantic interoperability’, you need matching impedance. You cannot (for example) have one system store an internal date of ‘1990’ as ‘1990-01-01 00:00:00’ and then emit this, to be taken as a precise date. But it’s much bigger than such trivial examples. I’ve looked around a bit for a FHIR ‘data model’ and come up with things like this from LeRoux et al.:
To my mind, structures like this have not been thought out adequately (even before you try to flesh out the details). To mention just a few problems with the above, it encourages episodic thinking without establishing problem lists and continuity over time; it is unscientific (in the sense I’ve already described ‘medical science’ previously); and documentation of interventions (and why they occurred) seems tenuous, implicit or absent. This is admittedly a specific instance (translation from ODM, which has its own issues) but I’ve struggled to find anything more solid.
Others, like those of Sarsipalle et al. are even less coherent:
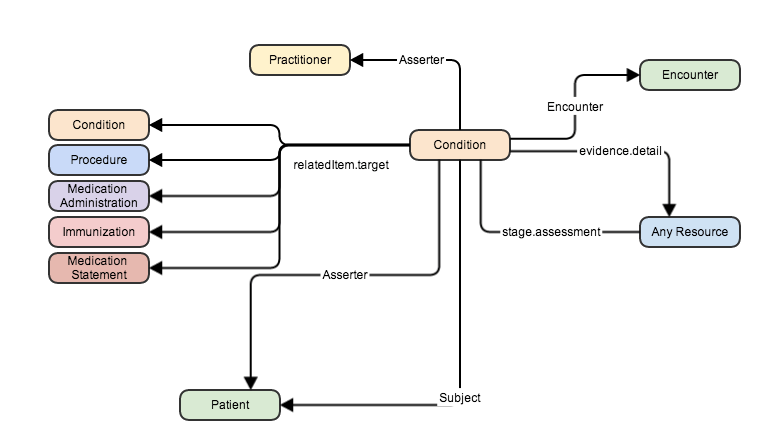
I’ve also seen David Hay’s attempts like this one:
Note that ‘Condition’ started out as ‘Problem’ but was renamed. Quite apart from the implication that either we have no problems, or that perhaps all conditions are problems, or vice versa (This can become rather messy), this is rather passive. How does a problem, uhh condition, prompt an action? Are two conditions causally related? Who owns which problems? What information supports/refutes the existence of the condition? And so on.
Perhaps I can summarise this all with a single question. The key question I’d like to ask another clinician about their actions is:
What were you thinking?
This and the associated information informs my subsequent decisions and actions. How does your model inform this question? I’d love to see it.
My 2c, Dr Jo.
Hira is developing an own entity model covering the health domain - I’ll see if there’s a version that can be posted here and invite them to join the discussion
Plenty to discuss in your latest post @DrJo , but I’ll start with your question on why HL7 International selected the term ‘Condition’ rather than ‘Problem’. Needless to say, there was a long debate about this, but the majority felt that Condition was a less pejorative term (e.g. describing ‘pregnancy’ as a problem doesn’t sit well with many pregnant women). SNOMED CT classifies ‘problem’ as a (primitive) clinical finding concept, whereas ‘condition’ is an attribute which supports clinical finding concepts such as ‘allergic condition’. From a consumer perspective, I don’t wish to see the term ‘Problem List’ on the health and wellness application of my choice.
Interestingly, (and given I am a man I may have this totally wrong) but classing pregnancy as a “condition” seems equally troubling for different reasons (ie. over medicalising). I will ask one of our midwifery advisors what she thinks.
Jon
…and I’ll ask the 2 midwives in my pub quiz team. ![]() Is the statement “a woman in your condition should (not) do xyz” not part of the Kiwi vernacular? As a matter of fact, ‘long term condition’ is a concept that’s captured in NZ primary care systems and, consequently, a member of the GP2GP Data Model and now an NZ FHIR Extension.
Is the statement “a woman in your condition should (not) do xyz” not part of the Kiwi vernacular? As a matter of fact, ‘long term condition’ is a concept that’s captured in NZ primary care systems and, consequently, a member of the GP2GP Data Model and now an NZ FHIR Extension.
Here is the response I got:
Hmmm…well it is a temporary condition but it’s not a pathological condition. ‘Diagnosis’ comes with negative connotations so let’s not call it that. We usually refer to pregnancy as a stage in the life course. I don’t suppose that helps at all???
Suggests, birth and death and maybe adulthood transition points might be different concepts to this (thinking about life course)…
Jon
FWIW, in the early days of the HL7 FHIR project, some of us proposed creating a Maternity Resource - but lost the relevant votes. HL7 International is a large, open, global and consensus-based organisation and abides by very strict ANSI rules (notably around handling dissenting views). It’s a very democratic process and, as with all democracies, not all decisions please everyone - but we have to live with them.
Allergies…intolerances…hypersensitivities - put any 3 SMEs in a room (e.g. at the 2016 SNOMED Expo in Wellington) and an implementer will hear 3 different opinions, none of which will probably match their previous understanding! Can a person be allergic to entire drug classes (e.g. penicillin) or food categories (e.g. seafood) - or, at the other extreme, must the exact substance(s) in question be identified? What supporting evidence is required (adverse reactions) and context (e.g. was the medication in question administered as instructed?). LSS - there is a reason why this domain is possibly the least well implemented in most EHRs.
As a rural GP, working with a population who have been dealt the worst hand of cards (e.g., negative social determinants of health), and so I am passionate about IT improving, not worsening, equity, and who reads all of the above with keen interest but mostly limited technical knowledge, my two cents are:
Any model should have whānau/people as the center of a model, if for no other reason, then to remind anyone using the model, why the model exists. Most definitely, this needs to be cyclical as @DrJo has highlighted.
Next, to be evidenced-based in an approach to improve equity, the foundation of a high quality health system that best achieves fair outcomes across a population is high quality primary care. [If anyone wants academic reading to back up this statement, let me know and I’ll add hyperlinks, etc.]. High quality primary care consists of being the first-point of contact to the health-system AND being accessible; providing comprehensive care; ensuring continuity-of-care; and, leading care coordination with the rest of the health system. These are B Starfield’s charactersitc, and again, I can link academic references if there is interest.
So, it does seem we need a strategic model to guide the rest of the operational and technical decisions that need to be made. How can such a strategic model be guided by evidenced-based models of care that improve equity? AKA, how can a strategic model be based on the principles of high quality primary care?
I tend to agree . . .
As a front-line condition, the foundational IT process that everything must support is a sophisticated patient portal. I am frustrated that patient portals seem to be an after-thought, rather than starting there, and ensuring all databases are built so that patient portals are the first user interface to test how the databases will be used. If we nail that, the user interface for clinicians, via EHRs, etc, will probably work better.
Finally, we have here in Aotearoa a wonderful opportunity to be informed by a non-English, and non-European linguistic history that absolutely can transform and improve strategic thinking. For example, the Hui Process (another academic reference, if there is interest), focuses on establishing the kaupapa for an episode-of-care. The concept of kaupapa, as I understand it as a non-Te Reo Māori speaker, seems to get closer to what’s helpful when talking to a patient: https://maoridictionary.co.nz/word/2439 than using terms like condition/problem which feel more rigid and less open to discussion and relationship-building . . . an integral part of healthcare.
@DrJo - I think that @emily.gill 's reply answers the question about hospital centricity better than I can. Her point about patient portals (preferably health & wellness apps chosen by the consumer, not one tethered to a provider’s PMS) is also highly significant if we are going to implement the patient-centric system/service laid out by the National Healthcare Strategy. In that respect care plans and care teams are critical entities, and I don’t believe that the example above (from LeRoux et al) does justice to that section of the FHIR Resource Model (e.g. it doesn’t link Patient to CarePlan), and I will produce a better version in the near future for the RCCC Project.
Hi @pkjordan
Well now my puzzled state has changed to bemusement. Let’s work through this.
-
You haven’t provided a single actual criticism of my model—is this death by implication? I note that you don’t have a model of your own yet, but would love to see how your model fits together when it appears.
-
The comments by @emily.gill struck me as pretty sensible. But in what way do they refute my model? Here’s what she said:
As a rural GP, working with a population who have been dealt the worst hand of cards (e.g., negative social determinants of health), and so I am passionate about IT improving, not worsening, equity
Agreed. Not a refutation of my model. My model is intended to facilitate just this.
Any model should have whānau/people as the center of a model, if for no other reason, then to remind anyone using the model, why the model exists. Most definitely, this needs to be cyclical as @DrJo has highlighted.
So @emily.gill agrees that the cyclical approach I’ve taken is solid. I’m not sure from her statement whether she is implying that my model is not patient-centric. It is. Every table relates directly to the patient, their problems, and their management. I thought this was pretty evident. If you’re interested in how best to implement this, then we can drill down to the details of table structures. Basically, I introduce an important denormalization that has several strengths—but as befits an introduction, I’ve kept things simple. Not a refutation, in any case. Happy to discuss details, if anyone is both interested and conversant with third normal form (and when to break it).
Next, to be evidenced-based in an approach to improve equity, the foundation of a high quality health system that best achieves fair outcomes across a population is high quality primary care. … High quality primary care consists of being the first-point of contact to the health-system AND being accessible; providing comprehensive care; ensuring continuity-of-care; and, leading care coordination with the rest of the health system.
Absolutely agree on all points. My model is designed to improve continuity of care—by extending its processes past the episodic ‘models’ we’ve seen to date. Continuity of care is ensured by ownership of problems—and hand over. But here we are starting to discuss deployment of my model—or any other model. Where can you see “bits that won’t work”? Again, I’m puzzled.
So, it does seem we need a strategic model to guide the rest of the operational and technical decisions that need to be made. How can such a strategic model be guided by evidenced-based models of care that improve equity? AKA, how can a strategic model be based on the principles of high quality primary care?
Yes, we need a model. We agree, again.
I think there may however be a bit of confusion here. If you have a robust schema, then it must be implementable in every health care setting. And—of course—it must be built in turn around a solid model of how medical science works. Mine is, to the best of my ability. Happy to discuss instantiation of the model, which is now what @emily.gill is talking about. Happy to discuss how it may be improved. Happy to put it up against any other models you have, or indeed might come up with. But I can’t see a refutation here. What did I miss?
I tend to agree . . .
As a front-line condition, the foundational IT process that everything must support is a sophisticated patient portal. I am frustrated that patient portals seem to be an after-thought, rather than starting there, and ensuring all databases are built so that patient portals are the first user interface to test how the databases will be used. If we nail that, the user interface for clinicians, via EHRs, etc, will probably work better.
Again we agree. I agree that current implementations have been woefully inadequate in addressing the problems that patients have. And indeed—I can’t recall a patient ever presenting with the statement “I have a condition. I’ve got this crushing chest pain going to my jaw”. Patients come to me with problems.* If we don’t allow patients to articulate their problems adequately—make this almost trivial to do—then we are doing them a disservice. Happy to explore implementation of my model in a primary care setting. Or indeed, any competing model—and it would be ideal to have open-source competition here, where models can be weighed against one another.
Patients also like to know what their clinicians are thinking. If even other clinicians can’t determine this from the record with any fidelity, where are we? Nowhere. One of the commonest things patients say to me is “I don’t get the impression my doctors are listening to me” (Two patients independently said this to me today). Perhaps the second commonest is “I don’t understand why this is being done”. Third on the list is “What’s going to happen next?” I’m sure that similar problems permeate our entire current health care system.
Which brings me to perhaps the most important point: one size does not fit all.
One size does not fit all
We need careful semantics here. You’ll recall that in a recent post, I said:
The ERD also doesn’t show a second-tier mapping that accommodates individual user groups and their representational needs.
I think this is a key issue where some of the pronouncements so far have generated more heat than light. Without diving too deep, let’s look at that pejorative word “problem” in more detail.
No. To a clinician, the term ‘problem’ is not pejorative. When they use the term, it has a functional connotation, and there is no adequate replacement, nor should there be. Science—and medical science—works with problem solving. As I’ve already intimated, patients quite often come to clinicians everywhere with problems. And we solve them. Provided we characterise them as such, and employ the mechanisms of science to address them.
To abandon problems is to abandon science. If you’re going to do that—well then good luck in managing your diabetes with the ear candling and crystal healing and Ayurvedic whatnot. This is where I bow out.
Nope. I’m not saying the patient view should be coerced into a mould that they find inimical or even slightly disrespectful. Quite the opposite. Lest you get the wrong message, let me illustrate this in more detail. If you’ve seen This is Going to Hurt, then you’ll almost certainly recall the senior clinicians being lectured in a patronisingly arrogant way by a clueless administrator about the terminology they must now use. The new PC terminology, for the week. They left the room. One size doesn’t fit all—and this cuts both ways.
More trenchantly I’m reminded of the following dialogue from the Hitch Hiker’s Guide to the Galaxy. Arthur the hero and his friend Ford are marooned on Earth with a enormous fleet of Golgafrinchan administrators, who are trying to plan their society:
CHAIRMAN: Yes, and, and, and the wheel. What about this wheel thingy? Sounds a terribly interesting project to me.
MARKETING GIRL: Er, yeah, well we’re having a little, er, difficulty here…
FORD: Difficulty?! It’s the single simplest machine in the entire universe!
MARKETING GIRL: Well alright mister wise guy, if you’re so clever you tell us what colour it should be!
Two levels
To me, this comically illustrates what happens when we confuse two levels:
- The modelling level, for which I’ve presented a basic, well, model. Call this “Tier I”.
- The deployment level, which—for the reasons illustrated—needs to provide different perspectives (and even subtly different terminology) for different people. Call this “Tier II”.
A key observation here is that we can actually model both levels—provided we implement solid mechanisms for (a) mapping between the two; and (b) limiting the damage when things go wrong. [Spoiler: my model also does this].
This not just for “consumers of health care”. It’s for everyone. The orthopaedic surgeon wants to see a different view from the geriatrician, who wants to see a different view from the physiotherapist. And Mr Henare, who is actually having the joint replacement, wants a very different view that makes complete sense to him. Yet we need a unified model underlying their different views. This is not easy, and it’s where most EHRs (and indeed, entire health systems) fall down.
We can get into a lot of trouble arguing about the colour of the wheel. We can also get into a huge crock of poo if we insist that those who are talking about Tier I need to use some alien Tier II term, simply because someone feels that this is the ‘right’ thing to do.
But all of this simply evaporates in the face of using the wrong terminology. If you didn’t click on the link that I inserted into a previous post—one that explains why it’s actually terminally daft to insist on saying ‘Condition’ when you mean ‘Problem’, here it is again.
In summary
Don’t confuse the modelling level with the way things are deployed. We need to:
- Get the basic model right (Tier I).
- Make the resulting front end optimally useful to everyone who uses the system. This involves modelling and implementing Tier II correctly—and sorting out the mapping between the two.
Having pettifogging arguments about what you should call your variables and table names on Tier I is not just silly. If we break the semantics because we want to be PC—or for any other reason—this can scupper our entire enterprise.
“When I use a word,’ Humpty Dumpty said in rather a scornful tone, ‘it means just what I choose it to mean — neither more nor less.
So what I’d like to see next in this discussion is:
- Actual criticism of my model. Happy to delve into the details.
- Competing models. This is surely more important. It’s quite likely that when people actually sit down and explain their own models rather than just obliquely sniping at mine, both will benefit—and we’ll make progress. If my model is crap—well, provide a better one. And let’s talk.
Let’s not spend our time arguing about the colour of the wheels.
My 2c, Dr Jo.
* I can almost guarantee that at least one person will misinterpret quite another ‘point of confusion’, so to pre-empt it, I feel obliged to insert this tiny footnote. Everything we’ve discussed so far pales into insignificance in the light of the societal determinants of healthcare. As we all know—or should know—population wellbeing depends primarily on how communities are dosed with commodities. It is the flow of commodities like alcohol and nicotine and sugar and high-energy-density foods and salt that largely determine community outcomes in terms of conditions like the biggest killer of all (hypertension: salt and inadequate provision of vegetables) as well as cancer (alcohol, tobacco), type 2 diabetes (crap food), chronic lung disease (tobacco) and so on.
With this preface, you can see that there are two distinct ways you can style ‘wellbeing’ and indeed the role of health care providers.
- We can act as advocates for the delivery of good health by limiting three things: the advertising, availability and price of these potentially harmful commodities. This is what we should all be doing—as any public health physician will tell you. Our powers here are almost completely limited to advocacy, because we have no other way we can influence the flow of those commodities. Politicians can, but they simply don’t.
- We can deceive ourselves into believing that we can control the flow of these commodities at a patient level by e.g. encouraging patients to “make the right choices”, or “concentrating on wellness”. This is, of course, risible.
I’m sure that someone on this list will accuse me of “focusing on problems” and thereby neglecting (2). Which is naturally absurd. As an advocate of (1), I’m happy to discuss how some otherwise smart clinicians have been taken in by (2).
@DrJo , I don’t claim to have an all-encompassing model of the healthcare domain - or wish to create one and evangelise it. I’ve seen far too many failed attempts to do that in my career. All I posted was that I could provide a more accurate representation of the FHIR Resource Model for Care Plans.
As an implementer, I’m happy to work with the FHIR Resource model because it is a solid basis for developing software for health information exchange projects. It neither is, or claims to be, perfect, but it’s the best that I’ve worked with.
From a consumer perspective, I have a number of issues with the way that healthcare data is provided and presented to me. I have long-term conditions that need to be managed by me with the occasional help of my GP (when she is available) and a wonderful osteopath. My GP encourages me to make the right choices with regard to exercise and diet and I am highly appreciative of this - I cannot believe that anyone would find that approach risible or foolish.
I was just in the process of writing you an email on this! Now that Hira, Primary care data programme and other special projects in the Gerard Keenan space are coming to fruition, how should information be modelled in a way that aligns both primary care, secondary care, patient/whanau and digital health industry (medtech, biotech, digital health/EMR) all align.
What standards within ISO/TC215 should be prioritised given it’s getting quite extensive on what can, should and must haves, and whether some of the current domestic developments supersede international needs?
For starters:
-
NZIPS covers what a patient is required for illness/medical health system, how does wellness health domain fit in?
-
Where does social determinants of health fall in from a data modelling?
-
Primary care data without notes and context is fairly bland without the provenance of how it was captured.
-
Same applies to device data whether it was a calibrated device or something that was expired or had poor sensitivity.
-
Hospital data definitions and interpretations vary, often driven by local strategy or contractual drivers. (e.g. 6 hour wait times in ED and how that’s measure inconsistently). How is that applied to the data model?
Just some food for thought.
Hi Samuel,
I don’t agree with your comment that Primary Care consultation level metadata is very bland. It depends on what your focus is. Currently, mine is population health, so the metadata plus dispensed medications seen through the lens of Information theory has a lot to offer. I would be happy and grateful for the metadata.
In a previous life, I audited GP notes. I don’t want them because of the variability. Some write “brief” notes, others use a confusing plethora of abbreviations, including me, and others write small novels. The latter group is also super-defensive about this information being released. I sympathise with their position and as I can’t think how to handle the variety anyway, cheerfully dodge the political angst.
Gathering the social determinants of health is a whole different challenge. That I don’t have a simple answer for.
Kind regards
Greig